Events Since the 20th Century:
2002-
The American Diabetes Association defines prediabetes as "impaired fasting glucose and/or impaired glucose tolerance." Impared fasting glucose is defined as a fasting blood glucose of 100-125 mg/dl, and impared glucose tolerance is defined as a glucose level from 140 mg/dl – 199 mg/dl two hours after consuming a set glucose amount.
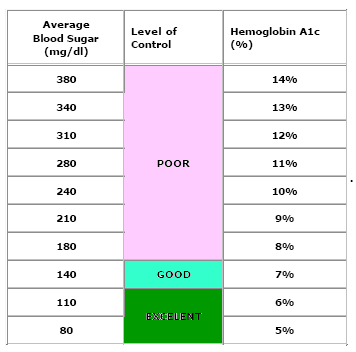
A1C levels of 5.7% to 6.4% are also used to identify individuals with pre-diabetes.
A1C levels of 5.7% to 6.4% are also used to identify individuals with pre-diabetes.
2003-
The fist tubeless insulin pump is approved (the omni pod). This remains the only tubeless pump on the market to this day.
2005-
Symlin, is approved in the U.S. as an injectable treatment for people who use insulin at mealtimes but still fails to achieve desirable blood glucose levels.
Byetta, is approved in the U.S. as a first-in-class incretin mimetic (GLP-1) drug to treat type 2 diabetes. An injectable drug, Byetta works by increasing insulin production in response to blood glucose levels.
Byetta, is approved in the U.S. as a first-in-class incretin mimetic (GLP-1) drug to treat type 2 diabetes. An injectable drug, Byetta works by increasing insulin production in response to blood glucose levels.
2013-
FDA approves Invokana, the first in a new class of drugs know as SGLT-2 inhibitors. These drugs lower elevated blood sugar in patients with type 2 diabetes. SGLT-2 inhibitors block the activity of sodium glucose transport proteins in the kidney, reducing glucose rises and increasing the removal of glucose in the urine.
2014-
Tandem creates T-slim the first touch screen insulin pump.